I 2 and I − complex to form the triiodide complex, which has a yellow color, unlike the white color of sodium iodide. Iodides (including sodium iodide) are detectably oxidized by atmospheric oxygen (O 2) to molecular iodine (I 2). Solubility of NaI (g NaI/kg of solvent at 25 ☌) Sodium iodide exhibits high solubility in some organic solvents, unlike sodium chloride or even bromide: After experiments on rabbits and dogs he settled upon sodium iodide as the best medium. 
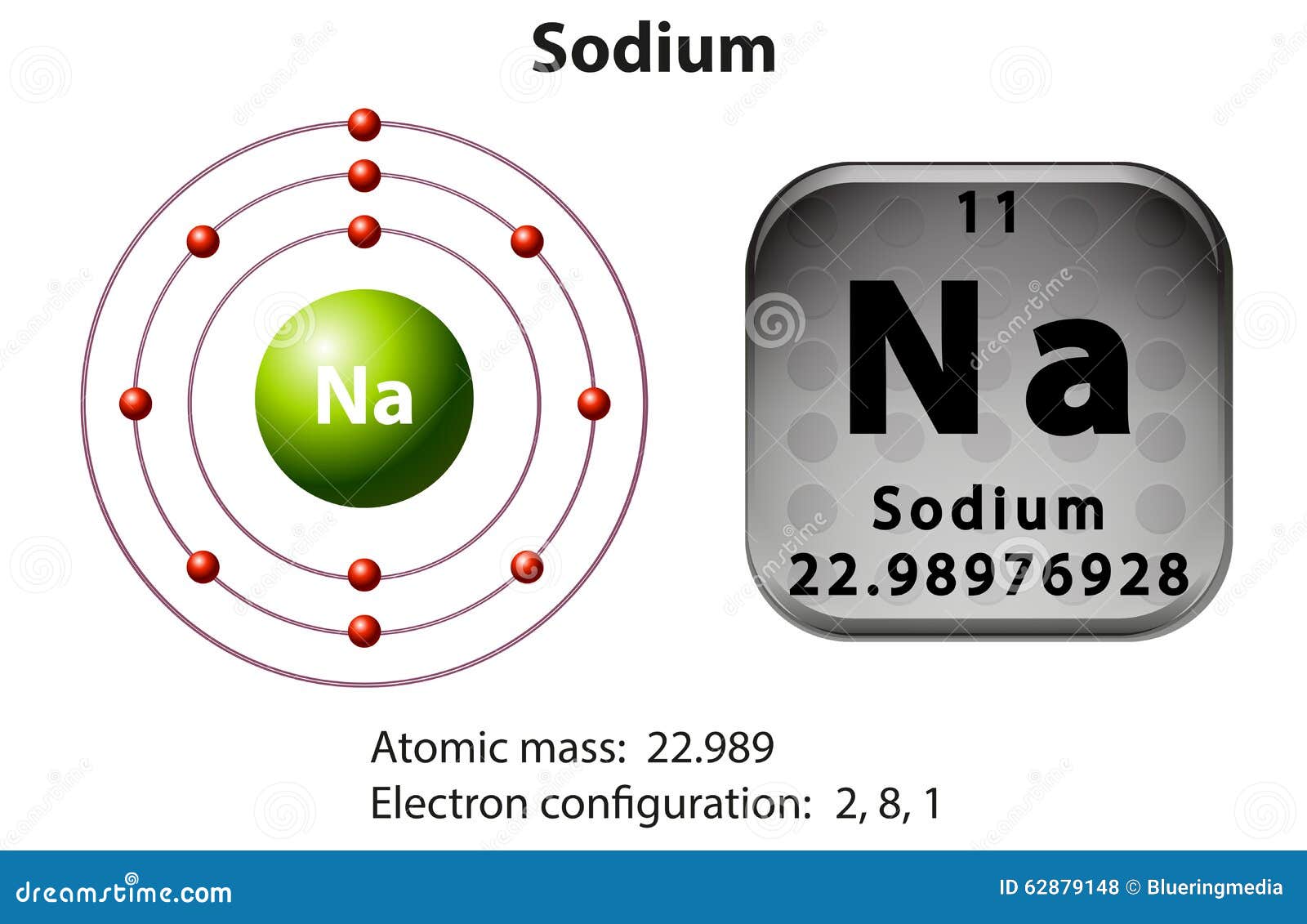
Radiocontrast Īntónio Egas Moniz searched for a radiocontrast agent for cerebral angiography. The wavelength of maximum emission is 415 nm. Sodium iodide can be used both as single crystals and as polycrystals for this purpose. Crystals with a higher level of doping are used in X-ray detectors with high spectrometric quality. Fine-tuning of some parameters (i.e., radiation hardness, afterglow, transparency) can be achieved by varying the conditions of the crystal growth. The crystals are usually coupled with a photomultiplier tube, in a hermetically sealed assembly, as sodium iodide is hygroscopic. NaI(Tl) is the most widely used scintillation material. Sodium iodide activated with thallium, NaI(Tl), when subjected to ionizing radiation, emits photons (i.e., scintillate) and is used in scintillation detectors, traditionally in nuclear medicine, geophysics, nuclear physics, and environmental measurements. Some radioactive iodide salts of sodium, including Na 125I and Na 131I, have radiopharmaceutical uses for thyroid cancer and hyperthyroidism or as radioactive tracer in imaging (see Isotopes of iodine > Radioiodines I-123, I-124, I-125, and I-131 in medicine and biology). This method, the Finkelstein reaction, relies on the insolubility of sodium chloride in acetone to drive the reaction: R–Cl + NaI → R–I + NaCl Nuclear medicine Sodium iodide is used for conversion of alkyl chlorides into alkyl iodides. Organic synthesis Monatomic NaI chains grown inside double-wall carbon nanotubes. Iodized table salt contains 10 ppm iodide. Sodium iodide, as well as potassium iodide, is commonly used to treat and prevent iodine deficiency. It is produced industrially as the salt formed when acidic iodides react with sodium hydroxide. It is used mainly as a nutritional supplement and in organic chemistry. Under standard conditions, it is a white, water-soluble solid comprising a 1:1 mix of sodium cations (Na +) and iodide anions (I −) in a crystal lattice. Sodium iodide ( chemical formula NaI) is an ionic compound formed from the chemical reaction of sodium metal and iodine. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
